 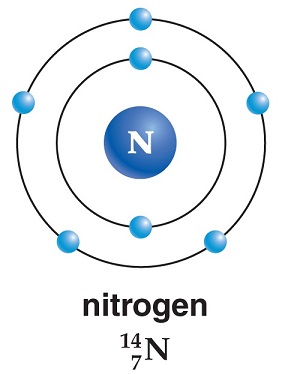
Online calculator, figures and tables showing density and specific weight of nitrogen, N 2, at temperatures ranging from -175 to 1325 C (-280 to 2400 F) at atmospheric and higher pressure - Imperial and SI Units. However, an element/compound in a solid state has a smaller state of entropy because molecules in a solid are less free to move. Nitrogen - Density and Specific Weight vs. The conclusion is reached that on the data, and employing van der Waals ' equation of state, the physical nature of Olszewski's experiment has still to be. The most stable of these structures has Nitrogen in the middle, double For each. Applying this to a chemistry example:CO 2 (g)-> CO 2 (s)An element/compound in a gaseous state always has a greater state of entropy (gaseous molecules are more free to move). N2O Lewis Structure, Resonance Structures, Oxidation Number. The state of disorder of things was lessened. The entropy of the universe always remains. It can either increase than the initial or decrease than the initial, but in an actual sense, entropy cannot decrease. For the change in entropy to be negative just think of it in terms of the room analogy: initially, it was messy, but then it got neater. Based on the increase or decrease in temperature, pressure or volume, entropy can change for a thermodynamic system, but the change can only be in accordance with the initial state. (For example, there are more ways to have a messy room than to have an impeccable, neat room). Remember entropy is essentially the state of disorder of a system since (on a macroscopic level) the natural progression of the world is from order to disorder. Since Boltzmann's constant is a positive number and ln(omega) will always be greater or equal to zero, entropy will never be negative.However, when calculating delta S (change in entropy in a thermodynamic process), yes entropy can be negative. Why? Omega (the number of microstates possible for a certain state) can never be smaller than one. So I'll answer for both.For S (entropy), which is defined by the function S=kln(omega), where k is Boltzmann's constant and omega is the number of microstates corresponding to a given state, the answer is no. I am not sure whether you refer to delta S (change in entropy) or entropy itself. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
